 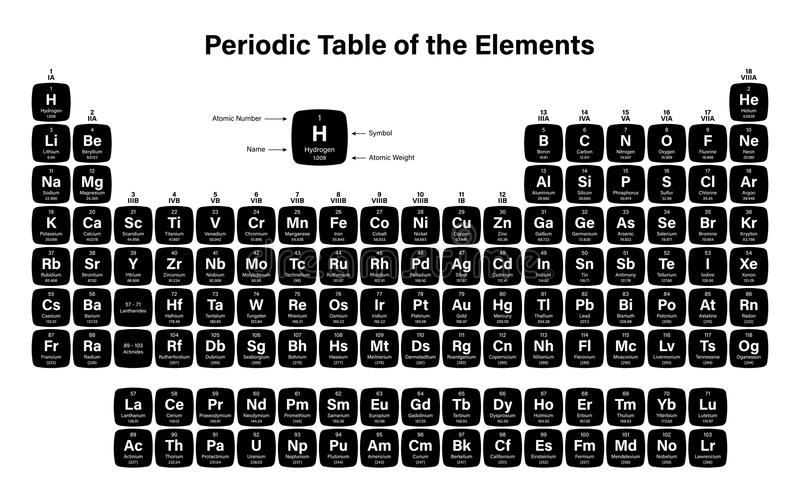
He proposed that "the properties of the periodic table of elements are the properties of numbers" as a result of the helical curve, which moved closely related elements into corresponding points above or below one another on the cylinder, a stunning prediction in light of present knowledge. de Chancourtois suggested an element classification based on Stanislao Cannizzaro's 1858 system's new atomic weight values. Later, attempts were made to show that the atomic weights Later, attempts were made to demonstrate that the atomic weights of the elements could be stated by an arithmetic function, and in 1862, A.-E.-B. One family includes oxygen, sulphur, selenium, and tellurium, while another includes nitrogen, phosphorus, arsenic, antimony, and bismuth. Fluorine was added to the halogens, and magnesium was added to the alkaline-earth metals, showing that similar relationships extended beyond the triads of elements. Döbereiner discovered that strontium's combining weight, or atomic weight, is midway between that of calcium and barium in 1817, and that other "triads" exist a few years later (bromine, iodine (halogens), lithium, potassium (alkali metals) sodium, and chlorin).īetween 18, J.-B.-A. In fact, even half a century after the systems of classification of compounds became widely used, chemists couldn't agree on how to classify elements. Relationships were more easily detected among compounds than among elements, resulting in an element classification that was several years behind that of compounds. This rapid expansion of chemical knowledge necessitated categorization, because not only the formalised literature of chemistry, but also the laboratory arts by which chemistry is passed down as a living science from one generation of chemists, are based on classification. Students can also avail the periodic table pdf from this article.Īnalytical chemistry-the art of distinguishing between different chemical substances-underwent rapid development in the early 19th century, resulting in the accumulation of a substantial body of knowledge about the chemical and physical properties of both elements and compounds. The periodic table of elements chart is represented below. The modern periodic table is also referred to as the dynamic periodic table or the periodic table with atomic mass. This clarification has increased the law's worth, and it is still in use now as much as it was at the turn of the 20th century, when it expressed the only known link between the elements. 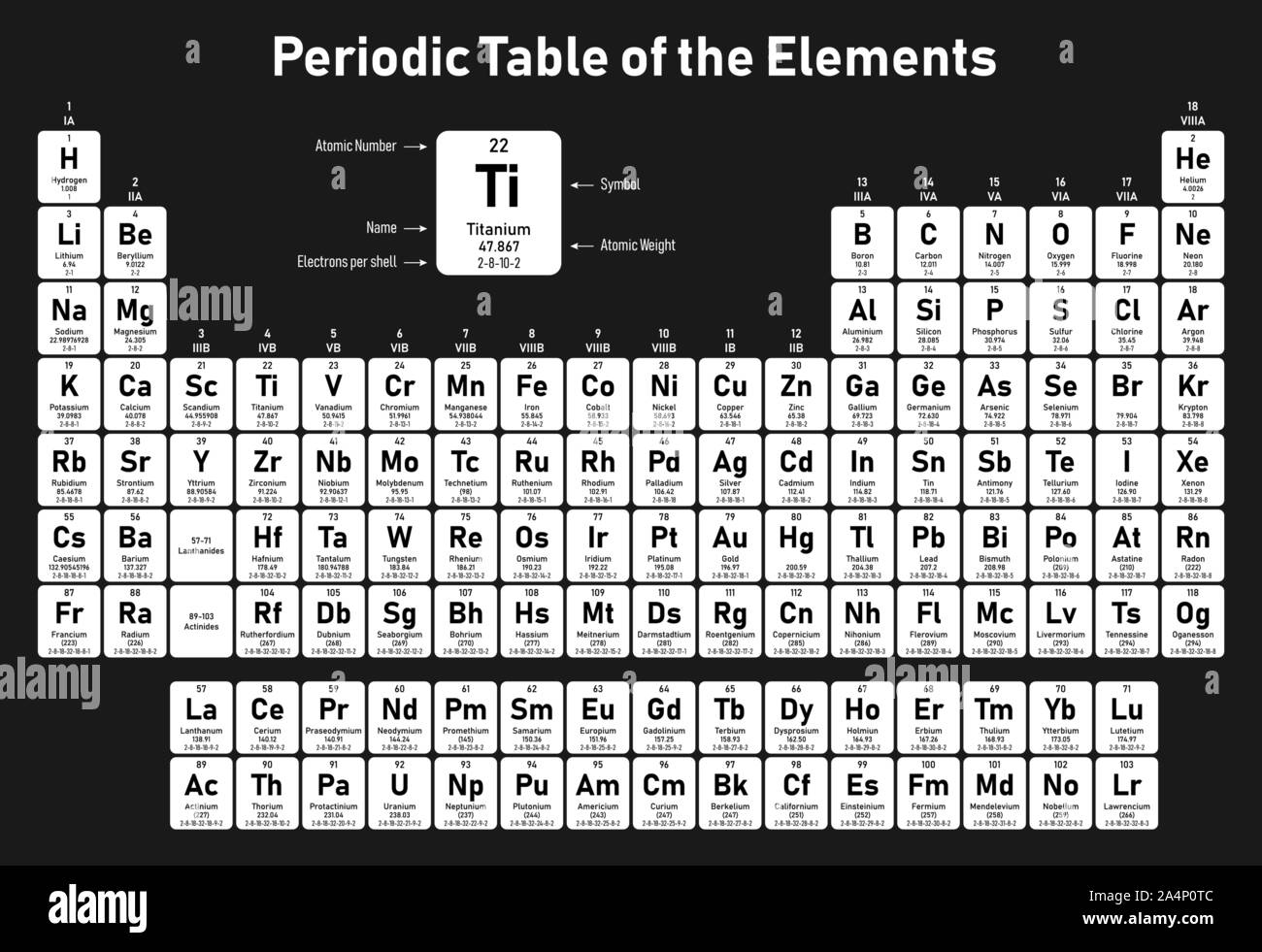
In the years since, researchers have made significant progress in explaining the periodic law in terms of atoms and molecules' electrical structure. This was not realised until the second decade of the 20th century. The order of elements in the periodic system is determined by their atomic numbers, the integers of which are equal to the positive electrical charges of the atomic nuclei represented in electronic units. Let us understand what is periodic table and more details associated with it from this article. The most reactive element in periodic table is Fluorine. Mendeleyev's first discovery, achieved in the mid-19th century, has been invaluable to the development of chemistry. When chemical elements are grouped in this way, their properties follow a recurring pattern known as the "periodic law," in which elements in the same column (group or periodic table groups) exhibit similar properties. The periodic table is an organised list of all chemical elements in order of increasing atomic number-that is, the total number of protons in the atomic nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
